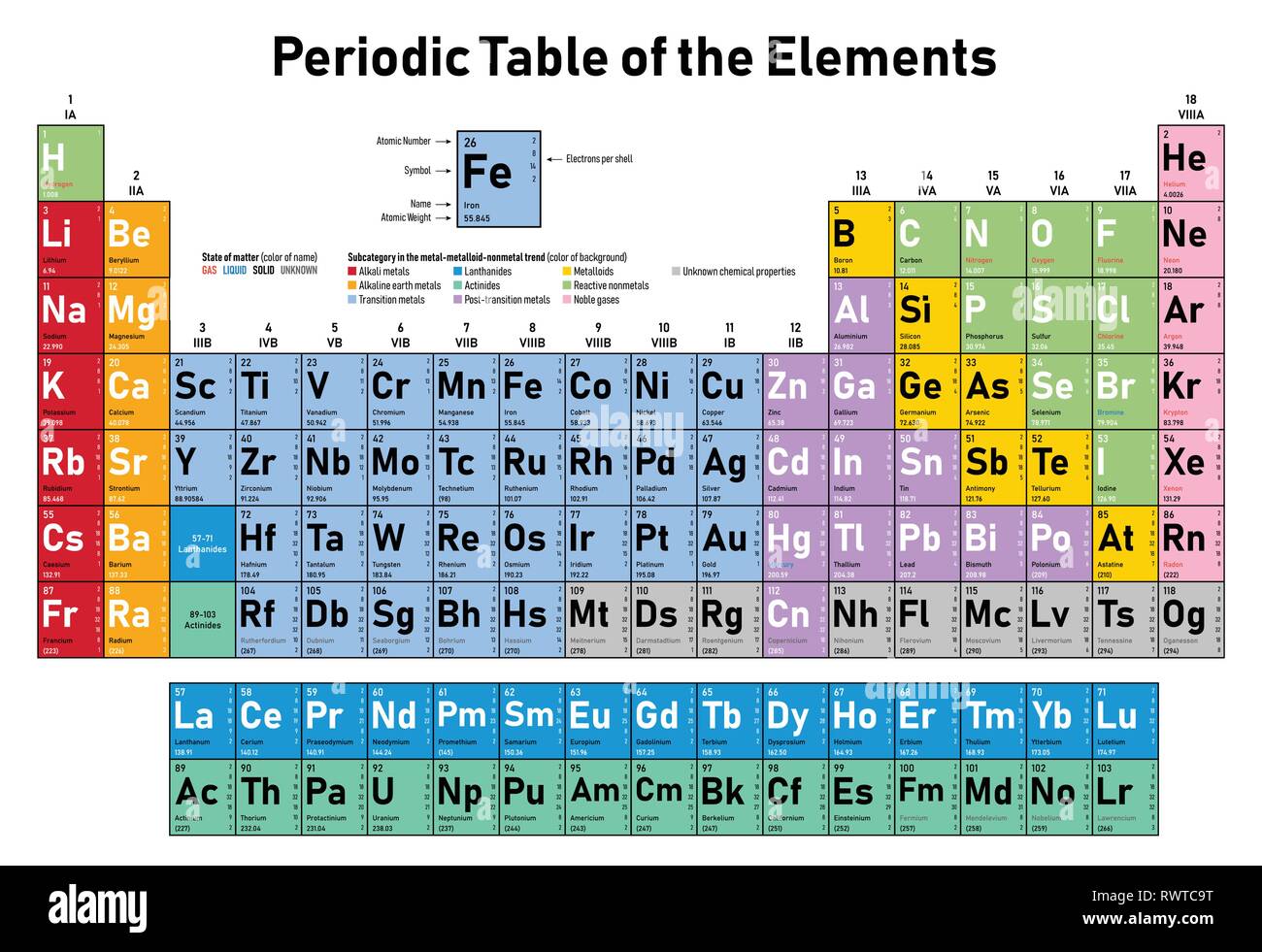
However, in consideration of the elements' observed chemical properties, he changed the order slightly and placed tellurium (atomic weight 127.6) ahead of iodine (atomic weight 126.9). Loosely speaking, the existence or construction of a periodic table of elements creates an ordering of the elements, and so they can be numbered in order.ĭmitri Mendeleev claimed that he arranged his first periodic tables (first published on March 6, 1869) in order of atomic weight ("Atomgewicht"). History The periodic table and a natural number for each element Russian chemist Dmitri Mendeleev, creator of the periodic table. 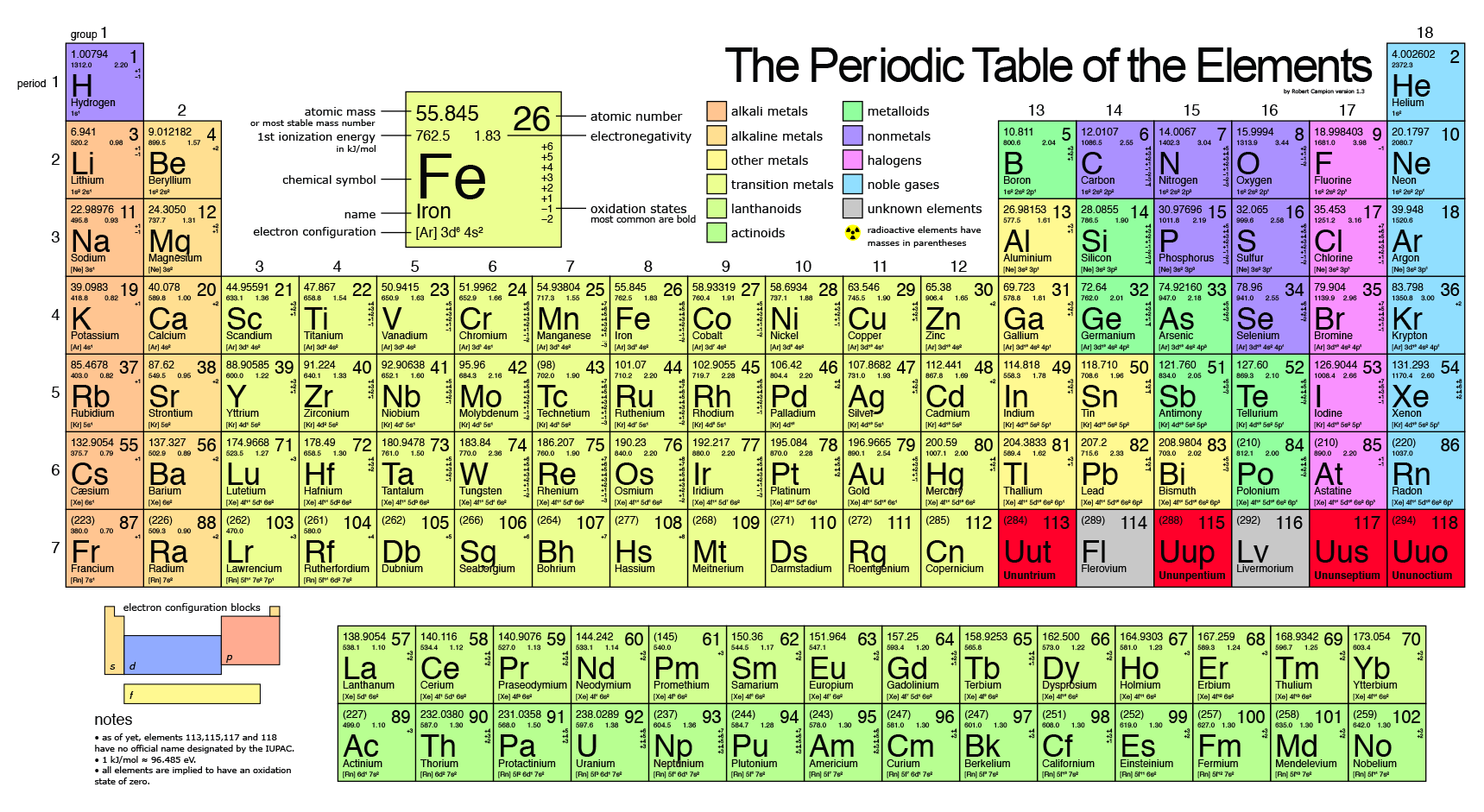
The rules above do not always apply to exotic atoms which contain short-lived elementary particles other than protons, neutrons and electrons. Only after 1915, with the suggestion and evidence that this Z number was also the nuclear charge and a physical characteristic of atoms, did the word Atom zahl (and its English equivalent atomic number) come into common use in this context. The conventional symbol Z comes from the German word Zahl 'number', which, before the modern synthesis of ideas from chemistry and physics, merely denoted an element's numerical place in the periodic table, whose order was then approximately, but not completely, consistent with the order of the elements by atomic weights. Historically, it was these atomic weights of elements (in comparison to hydrogen) that were the quantities measurable by chemists in the 19th century. A little more than three-quarters of naturally occurring elements exist as a mixture of isotopes (see monoisotopic elements), and the average isotopic mass of an isotopic mixture for an element (called the relative atomic mass) in a defined environment on Earth, determines the element's standard atomic weight. Since protons and neutrons have approximately the same mass (and the mass of the electrons is negligible for many purposes) and the mass defect of the nucleon binding is always small compared to the nucleon mass, the atomic mass of any atom, when expressed in daltons (making a quantity called the " relative isotopic mass"), is within 1% of the whole number A.Ītoms with the same atomic number but different neutron numbers, and hence different mass numbers, are known as isotopes. In an ordinary uncharged atom, the atomic number is also equal to the number of electrons.įor an ordinary atom which contains protons, neutrons and electrons, the sum of the atomic number Z and the neutron number N gives the atom's atomic mass number A. The atomic number can be used to uniquely identify ordinary chemical elements. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number ( n p) or the number of protons found in the nucleus of every atom of that element. The atomic number or nuclear charge number (symbol Z) of a chemical element is the charge number of an atomic nucleus. Both the concept of atomic number and the Bohr model were thereby given scientific credence. Experimental measurement by Henry Moseley of this radiation for many elements (from Z = 13 to 92) showed the results as predicted by Bohr. In this model it is an essential feature that the photon energy (or frequency) of the electromagnetic radiation emitted (shown) when an electron jumps from one orbital to another be proportional to the mathematical square of atomic charge ( Z 2). The Rutherford–Bohr model of the hydrogen atom ( Z = 1) or a hydrogen-like ion ( Z > 1). Atomic number is the number of protons, and therefore also the total positive charge, in the atomic nucleus. Not to be confused with Atomic mass, Mass number, or Atomic weight.Īn explanation of the superscripts and subscripts seen in atomic number notation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
